What is DURC?
Dual Use Research of Concern (DURC), under the United States Government Policy, is life sciences research that, based on current understanding, can be reasonably anticipated to provide knowledge, information, products, or technologies that could be directly misapplied to pose a significant threat, with broad potential consequences, to public health and safety, agricultural crops and other plants, animals, the environment, materiel, or national security.
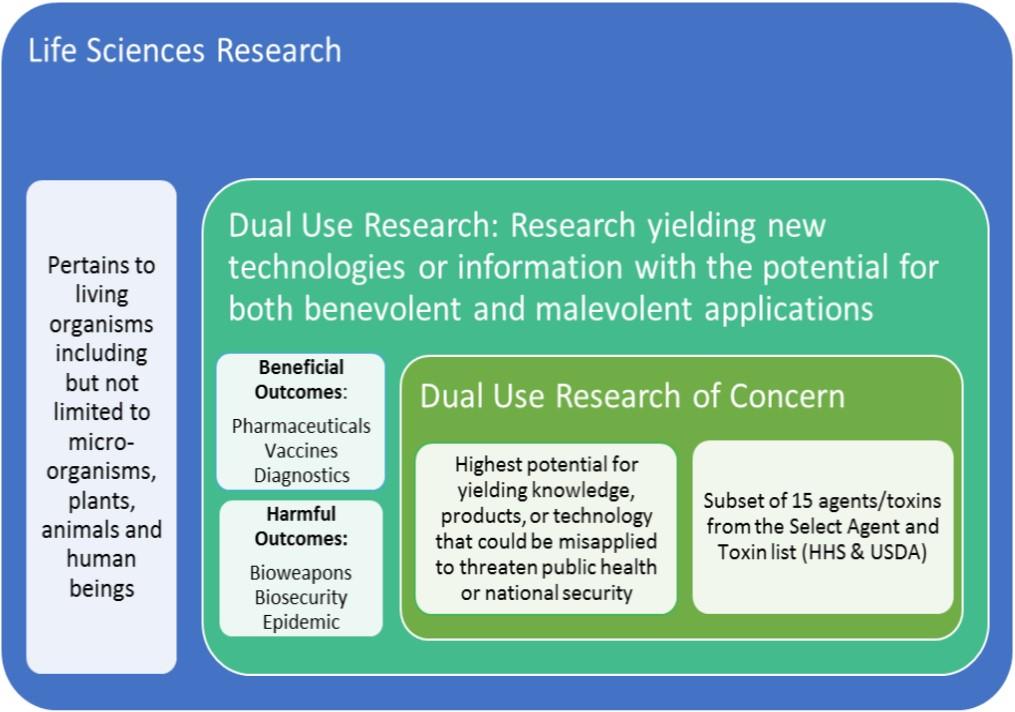
Dual Use Research vs Dual Use Research of Concern
Research yielding new technologies or information with the potential for both benevolent and malevolent applications is referred to as "dual use research." Some degree of dual use potential may be inherent in a significant portion of life sciences research. However, the small subset of life sciences research with the highest potential for yielding knowledge, products, or technology that could be misapplied to threaten public health or national security is referred to as "dual use research of concern."