COI Disclosure Requirements
The Federal and State governments and the University of California require financial disclosures from researchers for certain types of projects (see Forms & Policies). Principal Investigators on sponsored research agreements and Lead Researchers on IRB protocols have additional disclosure responsibilities, including ensuring that their research personnel comply with the conflict of interest policies.
All Researchers
KR Federal Disclosure and Certification
Who: Researchers on Public Health Service, Department of Energy, National Science Foundation, National Aeronautics and Space Administration, US Department of Agriculture compliant proposals and awards (see Forms & Policies for list of compliant organizations)
What: Create and maintain an updated KR Federal Disclosure and Certification (three yes/no questions). Third question relates to Malign Foreign Talent Recruitment Programs (see Foreign Talent Recruitment Program for more information).
For researchers with significant financial interests, additional disclosure forms may be required. The COI Team will notify the researcher directly for the additional forms.
- PHS/DOE/USDA: Annual Form 810, Form 820
- NSF/NASA: Federal Supplemental Disclosure to determine if have a related NSF/NASA significant financial interest, COI Addendum (COI-1)
For researchers that are a party to a malign foreign talent recruitment program or do not know if they are, they will be referred to the Research Security & International Engagement (RSIE) Team for additional guidance.
How: KR Federal Disclosure and Certification
More information:
ZotIRB Application
Who: Researchers engaged in human subjects research
What: Need to notify the Lead Researcher if you have a disclosable financial interest related to the IRB protocol prior to the submission of the new IRB applications.
How: For new IRB protocol applications, Lead Researcher or administrative contact will check "yes" to the conflict of interest question in the ZotIRB application for the appropriate researchers.
Definitions:
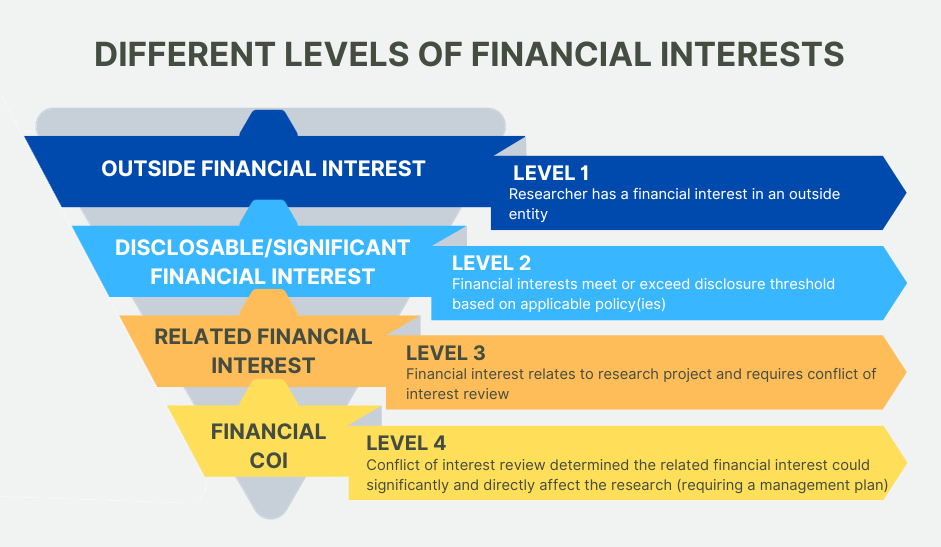
- Disclosable financial interest: The researcher (or researcher’s spouse/registered domestic partner or dependent children) have:
- Outside income equal to or exceeding $10,000 over the preceding 12 months (includes salary, consultant payments, honoraria, royalty payments, dividends, loan or any other payments or consideration with value, including payments made the University Health Sciences Compensation Plan);
- Equity (including stock, stock options, or any other investment or ownership interest) in a publicly held company equal to or exceeding $10,000 (current market value) or a 5% or greater ownership interest;
- Any equity (including stock, stock options, or any other investment or ownership interest) in a privately held company, regardless of value;
- Any management position, such as Board of Directors, director, officer, partner, or trustee; and/or
- Intellectual property interest, e.g., a patent (actual, planned, or applied for) or a copyright for software assigned or to be assigned to a party other than the UC Regents.
- Related financial interests: Related financial interests occur when the researcher, their spouse/registered domestic partner or dependent children have a disclosable financial interest that would reasonably appear to be affected by the research or when the entity in which the financial interests are held would reasonably appear to be affected by the research.
New or changed significant financial interest disclosure
Who: Investigator on a PHS/DOE/USDA or NSF/NASA compliant study or research personnel on an IRB protocol
What: The above listed individuals are required to report acquiring or discovering a new significant financial interest (PHS/DOE/NSF/NASA/USDA) or related disclosable financial interest (IRB)
How: The Disclosing Individual should email coioc@research.uci.edu to notify the COI Team of the new significant/disclosable financial interest for instructions.
More information:
Related Publications, Presentations, and IRB Consents
Who: Disclosing Individual (researcher reviewed by the Conflict of Interest Oversight Committee)
What: The Disclosing Individual is required to disclose their related financial interests in presentations, publications, and if applicable, informed consent for human subjects research. This has long been a standard COI Best Practices Requirement for all disclosures reviewed by the COIOC.
How: The Disclosing Individual include disclosures in presentations, publications, and if applicable, informed consent for human subjects research
Study/Lab Team Disclosure (COI Point Person)
Who: Disclosing Individual (researcher reviewed by the Conflict of Interest Oversight Committee) on new awards
What: The Disclosing Individual is required to disclose their financial interests to the study team working on a project related to their financial interests and identify a non-conflicted, non-junior person to contact if they have any COI questions or concerns ("COI Point Person"). No action required by the non-conflicted study team members other than raising COI concerns and questions to the COI Point Person.
How: The Disclosing Individual will email the study team members, the COI Point Person, and coioc@research.uci.edu a description of the financial interest and the identity of the COI Point Person
Other Relevant Policies
- Research Security & International Engagement
- Students & Conflict of Interest
- Conflict of Commitment (Faculty)- UC OATS
- UC Patent Policy- All inventions created by UCI employees during their UCI period of employment need to be disclosed to the Research Translation Group, including any inventions generated by UCI employees under a consulting agreement with an outside entity. Contact the RTG team at rtg@uci.edu.
Principal Investigators/Lead Researchers
Form 700U (non-governmental sponsor)
Who: Principal Investigators and Co-Principal Investigators on non-governmental funded research grants, contracts, gifts, and some incoming material transfer agreements (see list of exemptions below)
What: Disclose financial interests in the non-governmental sponsor/donor
How: Submit a Docusigned copy of the Form 700U
- Principal Investigator Only Form 700U (for cases when the PI completes the entire form)
- In the DocuSign PowerForm Signer Information page, enter the PI's name and UCI email address in the first two text boxes.
- (Optional) Enter a Department Administrator name and UCI email address in the last two text boxes if they should also receive a copy of the completed Form 700U
- In the DocuSign PowerForm Signer Information page, enter the PI's name and UCI email address in the first two text boxes.
- Department Administrator/Delegate and PI Form 700U (for cases when a department administrator or other delegate will complete the first sections of the form before sending it to the PI to complete the financial interest section and sign)
- In the DocuSign PowerForm Signer Information page, enter the Department Administrator’s name and UCI email address in the first two text boxes and enter the PI’s name and UCI email address in the last two text boxes.
- After the Department Administrator completes the top portion and sections 1 & 2, the form will route to the PI to complete and sign the Form 700U.
- Form 700U PDF- still needs DocuSign signature
More Information:
ZotIRB
Who: Lead Researchers
What: Ensure all research personnel engaged in human subjects research understand the IRB COI disclosure requirements, have disclosed any related financial interests in the IRB applications, and notify the COI Team when they have acquired or discovered a new related disclosable financial interest
How:
- For new IRB studies, indicate in the ZotIRB application which research personnel members have a disclosable financial interest related to the study.
- For a study team member reporting a newly acquired or discovered disclosable financial interests, submit an ZotIRB amendment to initiate the process.
KR Proposal Development (PHS, DOE, NSF, NASA funded)
Who: Principal Investigators on PHS, DOE, NSF, and NASA compliant proposals and awards
What: Principal Investigators must list all individuals involved in the project who meet the definition of an "Investigator", a person responsible for the design, conduct, or reporting of the project, in KR Proposal Development as Key Personnel. Ensure the Investigators understand their disclosure requirements including the requirement to update their disclosure if they have a new significant financial interests
Notify Sponsored Projects Officer if the list of Investigators needs to be updated during the period of the award.
How:
- New Proposal: KR Proposal Development
- Ongoing Award: Notify the C&G Officer if someone needs to be added or removed from the list of Investigators
- New Significant Financial Interest: Notify the COI Team, coioc@research.uci.edu
More Information: